 
Other important groupings of elements in the periodic table are the main group elements, the transition metals, the lanthanides, and the actinides. Metals are lustrous, good conductors of electricity, and readily shaped (they are ductile and malleable), whereas solid nonmetals are generally brittle and poor electrical conductors. They are separated by a diagonal band of semimetals. As of January 2016, the seventh row of the periodic table was completed with the addition of four. Metals are located on the left of the periodic table, and nonmetals are located on the upper right. A common answer among the students was to place vibranium near the bottom of the periodic table. Titanium, iron, and uranium are not representative elements. 
Semimetals exhibit properties intermediate between those of metals and nonmetals. Hydrogen, magnesium, gallium, and iodine are all representative elements. The elements can be broadly divided into metals, nonmetals, and semimetals. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. These metals are characterized by their soft texture and silvery color. They tend to donate their electrons in reactions and have an oxidation state of +1. This is due in part to their larger atomic radii and low ionization energies. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. Alkali metals are among the most reactive metals. It arranges of the elements in order of increasing atomic number. Metal reactivity relates to ability to lose. Four major factors affect reactivity of metals: nuclear charge, atomic radius, shielding effect and sublevel arrangement (of electrons). The periodic table is used as a predictive tool. The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. (group 2, or alkaline earth metals) of the periodic table: all group 2 elements have the. 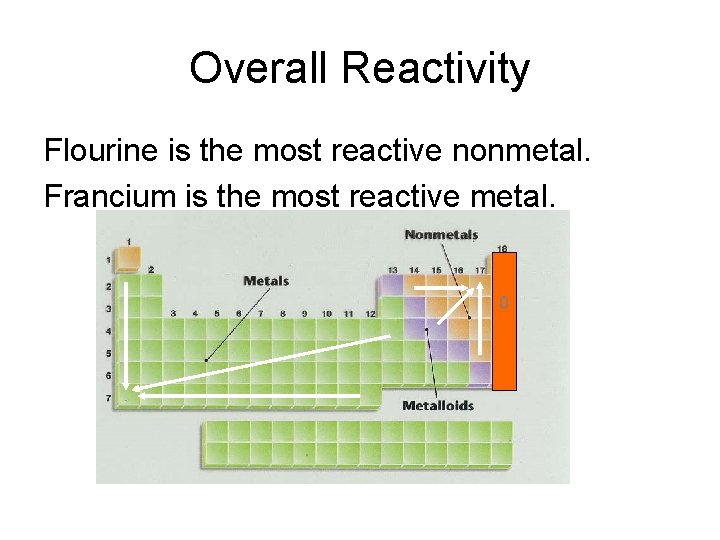
Sodium is an alkali metal, being in group 1 of the periodic table, because it has a single electron in its outer shell that it readily donates, creating a positively charged atomthe Na+ cation. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Sodium is a soft, silvery-white, highly reactive metal. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. This above image clearly shows you where are Alkali metals located on the Periodic table. Alkali metals include: Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs) and Francium (Fr). 1, reacts readily with metals to form compounds that can be broadly classified as salts and, therefore, are known as the halogens, which is derived from a combination of Greek words that translate to 'salt makers. Alkali metals are located in group 1 on the left side of the Periodic table. The periodic table is being read like a book: Elements are listed starting from the top, far left element (hydrogen) across the whole row before. The vast majority of the known elements are metals. Each of the elements found in this column, which are boxed in lavender in Figure 2.2.1 2.2. List the first 12 elements mentioned in the song. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. 
Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \( \newcommand\): Metal ions in a sea of electrons.\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
